
The Importance of Enzyme Immobilization
A key enabler to bring biocatalysis to scale.
Enzyme immobilization has been around for more than 100 years, and it keeps growing into a more and more essential technology in the processing industry. The reason for this is that immobilized enzymes can be deployed in a variety of applications, demonstrating high versatility. Over recent years, enzyme immobilization has constantly been further developed, making it simpler, quicker, and more efficient. - This just made it evermore appealing for practical implementation. Keeping this in mind, let’s take a closer look into the essentials and the purpose of enzyme immobilization.
What are immobilized enzymes?
The term ‘immobilized enzymes’ refers to heterogeneous, insolubile biocatalyst preparations. These can be prepared by a range of different strategies including binding of enzymes onto a solid support, entrapping enzymes in gels or microcapsules, or cross-linking enzymes to a degree at which they become insoluble. For more be referred to our Introduction to Enzyme Immobilization. The immobilization on solid supports is greatly enabled by a flurry of different available commercial enzyme carrier supports and thus a widespread approach.
Classically, enzyme immobilization can significantly increase enzyme stability and resistance to external factors such as temperature and pH. Moreover, comparing free and immobilized enzymes it becomes obvious that free, soluble enzyme will just be homogeneously distributed in the volume flow of the process, whereas immobilized enzymes remain concentrated and stationary in the compartment deployed. This renders separation and reuse of the biocatalyst much easier. However, the quality of immobilized enzymes is greatly influenced by immobilization carrier/support and immobilization process.
The purpose of immobilization
Just as for heterogeneous chemical catalysts, immobilized enzymes are easy to recover and re-use in both batch and continuous reactor operation modes. As also pointed out by S. Cantone et al. (2013), immobilization might also serve other purposes.
First, to avoid aggregation, precipitation or denaturation of free enzymes in organic solvents, their immobilization might pose an effective solution. Second, the use of immobilized enzymes allows to create localized reactor compartments with high catalyst-to-substrate ratio. Third, successful immobilization can provide stabilization required for long-term operation in an industrial setting. Lastly, in a few instances, immobilization may also increase enzyme activity.
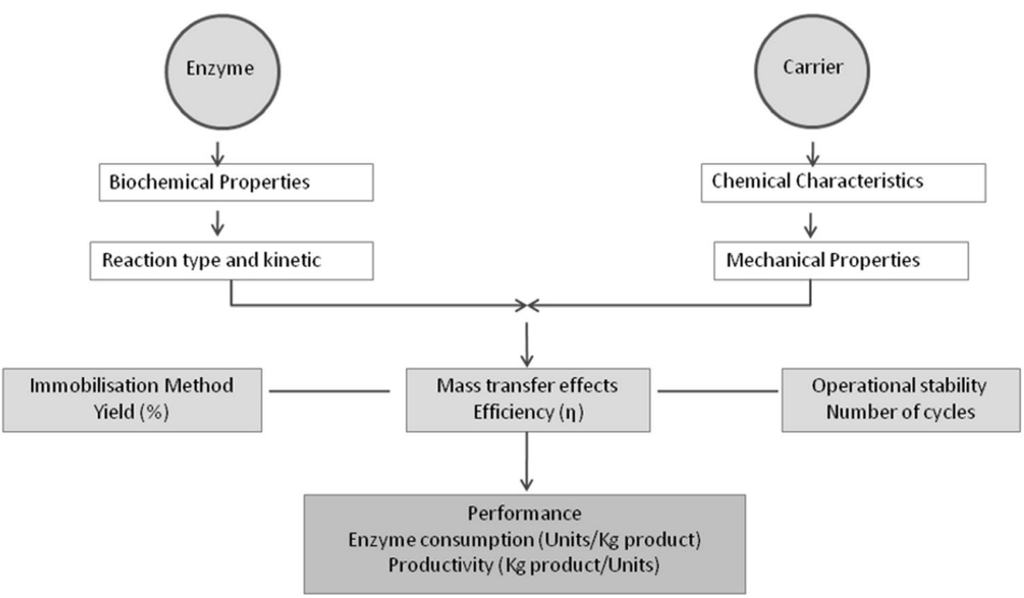
In an industrial setting, the ability to reuse the enzyme is especially important. The cost of the enzyme is typically a significant portion of the operating costs of a biocatalytic process and an added upstream immobilization step must pay off by enabling biocatalyst recycling. In this way, the total initial biocatalyst costs can be recouped.
Main reasons to opt for immobilization
- improve total catalytic productivity of enzymes
- reuse of enzymes
- simplified processes requiring less labor input
- overall reduction of cost
- reduced risk of product contamination
- stability of the product
- better process control
- high enzyme-to-substrate ratio
Contact us today to make your immobilization processes simpler, quicker, and more efficient!
References:
[1] S. Cantone, et al., Chem. Soc. Rev., 2013, 42(15), pp. 6262-6276. (image credit)


